Quick Overview
Understanding vapour pressure is essential for engineers working with industrial valves, pipelines, and fluid systems. Vapour pressure determines how easily a liquid evaporates and directly influences phenomena such as cavitation, boiling, and system stability. In industrial processes, ignoring vapour pressure can lead to equipment damage, safety risks, and costly downtime. This guide from Vcore Valve explains the concept, its influencing factors, and how engineers can manage it effectively in valve and piping applications.
A Quick Engineering Scenario
Imagine a process engineer reviewing a pipeline system transporting hot hydrocarbons.
The pressure drops suddenly across a control valve, and moments later the system begins vibrating loudly.
Within weeks, the valve trim shows pitting damage.
The culprit?
Cavitation caused by vapour pressure miscalculations.
Many industrial failures trace back to a simple misunderstanding: how liquids behave when pressure and temperature change.
Common Problems Engineers Face
1. Unexpected Cavitation in Control Valves
When system pressure drops below a fluid’s vapour pressure, bubbles form and collapse violently, damaging valve internals.
2. Incorrect Valve Selection
Ignoring vapour pressure characteristics can result in valves that cannot withstand flashing or high vapour formation.
3. System Instability in High-Temperature Processes
Fluids with high vapour pressure can transition to vapour rapidly, creating two-phase flow conditions.
Practical Solutions Engineers Should Apply
1. Calculate Vapour Pressure at Operating Temperature
Always evaluate vapour pressure vs operating temperature rather than relying on standard tables.
2. Select Cavitation-Resistant Valve Designs
Industrial valves should include:
-
multi-stage pressure reduction
-
anti-cavitation trims
-
hardened internal components
3. Maintain Safe Pressure Margins
Engineers typically maintain a pressure margin above vapour pressure to prevent flashing.
What Is Vapour Pressure?
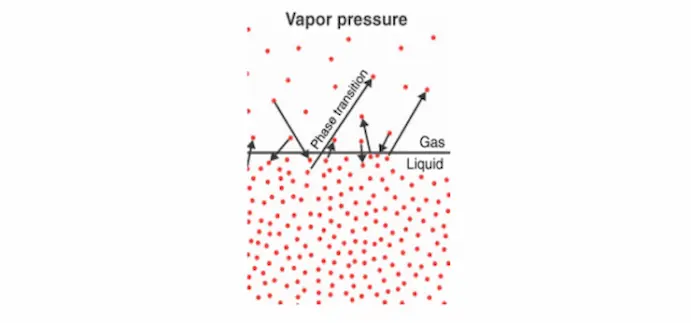
Vapour pressure is the pressure exerted by a vapour when it is in equilibrium with its liquid phase in a closed system.
In simple terms, it measures how easily a liquid turns into gas.
When molecules escape from the liquid surface and become vapour, they create pressure in the surrounding space. The balance between evaporation and condensation determines the vapour pressure.
A higher vapour pressure means a liquid evaporates more easily, making it more volatile.
Key Factors That Influence Vapour Pressure
Temperature
Temperature is the most important factor affecting vapour pressure.
As temperature increases:
-
molecular energy increases
-
more molecules escape the liquid
-
vapour pressure rises rapidly
Intermolecular Forces
Liquids with strong intermolecular forces (like water) have lower vapour pressure.
Liquids with weak forces (such as solvents) evaporate more easily.
Fluid Composition
Mixtures can have very different vapour pressure behaviour compared with pure fluids.
This is why chemical process engineering requires detailed thermodynamic modelling.
Vapour Pressure and Boiling Point
Boiling occurs when a liquid’s vapour pressure equals the surrounding pressure.
For example:
-
Water boils at 100°C at atmospheric pressure
-
At lower pressure (high altitude), boiling occurs at lower temperature
This same principle applies inside industrial pipelines.
If local pressure falls below vapour pressure, flash boiling can occur instantly.
Why Vapour Pressure Matters in Industrial Valve Systems
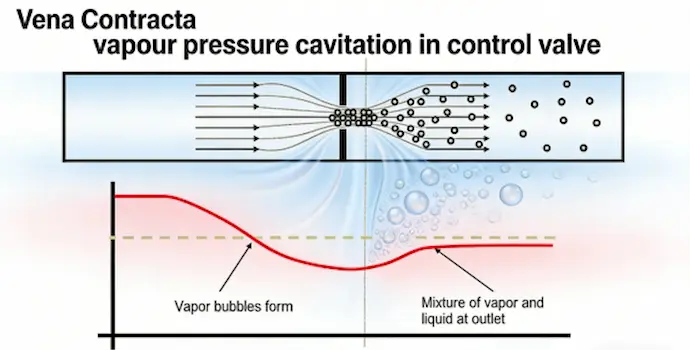
Cavitation
One of the biggest threats to valve performance is cavitation.
Cavitation happens when:
-
Fluid pressure drops below vapour pressure
-
Vapour bubbles form
-
Bubbles collapse when pressure rises
These implosions create shock waves that damage valve components.
Flashing
Unlike cavitation, flashing occurs when vapour bubbles do not collapse and remain in the flow.
This leads to:
-
erosion
-
vibration
-
reduced flow control accuracy
Fluid Selection
When selecting fluids or designing systems, engineers must evaluate:
-
vapour pressure
-
operating temperature
-
pressure drops across valves
Ignoring these factors can drastically shorten equipment lifespan.
Case Study: Cavitation in a Control Valve
A refinery installed a new flow control valve handling hot condensate.
Operating conditions:
Temperature: 160°C
Pressure drop: 12 bar
Within three months, engineers observed:
-
severe noise
-
vibration
-
internal valve damage
After analysis, the issue was identified as vapour pressure induced cavitation.
Solution implemented:
-
anti-cavitation valve trim
-
multi-stage pressure reduction
Result:
Valve lifespan increased over 4×.
Data Analysis
Below is a simplified comparison of vapour pressure values at 25°C.
| Substance | Vapour Pressure (atm) | Volatility Level | Industrial Impact |
|---|---|---|---|
| Diethyl ether | 0.7 | Very high | High evaporation risk |
| Bromine | 0.3 | High | Requires sealed systems |
| Ethanol | 0.08 | Moderate | Common process fluid |
| Water | 0.03 | Low | Stable fluid behaviour |
| Glycerol | extremely low | Very low | Minimal evaporation |
Higher vapour pressure indicates a greater tendency for liquids to evaporate.
For valve engineering, this directly affects cavitation risk and system stability.
Market and Engineering Trends
Industrial process industries are increasingly focused on fluid dynamic optimisation.
Emerging trends include:
-
AI-based flow simulations
-
predictive cavitation modelling
-
smart valve monitoring systems
Modern valve manufacturers like Vcore Valve are integrating advanced trim designs to mitigate vapour-related damage.
As industries demand higher efficiency, understanding thermodynamic properties like vapour pressure becomes even more critical.
Engineering Tips for Managing Vapour Pressure
Engineers can reduce vapour-related problems by following several key practices.
1. Avoid excessive pressure drops
Design piping systems to minimise sudden pressure reductions.
2. Use staged pressure control
Multi-stage valves reduce cavitation risk.
3. Monitor process temperature
Temperature increases can dramatically increase vapour pressure.
4. Choose appropriate valve types
Globe valves with anti-cavitation trims often perform better in these conditions.
Conclusion
Understanding vapour pressure is essential for designing safe and reliable industrial fluid systems.
From cavitation prevention to fluid selection and valve design, vapour pressure plays a critical role in engineering performance.
By considering vapour pressure during system design and selecting appropriate equipment, engineers can avoid costly failures and improve long-term reliability.
At Vcore Valve, we help industrial operators choose the right valves for demanding fluid conditions, ensuring stable operation and extended equipment life.
FAQs
What is vapour pressure in simple terms?
Vapour pressure is the pressure created by a liquid’s vapour when evaporation and condensation reach equilibrium.
Why is vapour pressure important in valves?
It determines whether cavitation or flashing may occur inside valves.
Does vapour pressure increase with temperature?
Yes. Higher temperature gives molecules more energy to escape the liquid phase.
What happens if pressure falls below vapour pressure?
The liquid begins to vaporise, forming bubbles that may lead to cavitation.
Can vapour pressure damage industrial valves?
Yes. Cavitation caused by vapour pressure differences can erode valve trims and seats.
How do engineers prevent cavitation?
By maintaining sufficient pressure margins and using anti-cavitation valve designs.
Reference
Britannica – Vapour Pressure Definition
https://www.britannica.com/science/vapor-pressure
Purdue University – Vapour Pressure Data
https://www.chem.purdue.edu/gchelp/liquids/vpress.html
US Geological Survey – Vapour Pressure and Water
https://www.usgs.gov/special-topics/water-science-school/science/vapor-pressure-and-water
AIChE Process Safety Glossary – Vapor Pressure
https://www.aiche.org/ccps/resources/glossary/process-safety-glossary/vapor-pressure